New oral anticoagulants (NOACs) are skyrocketing in popularity. Here’s what you need to know about this target-specific reversal agent for Dabigatran.
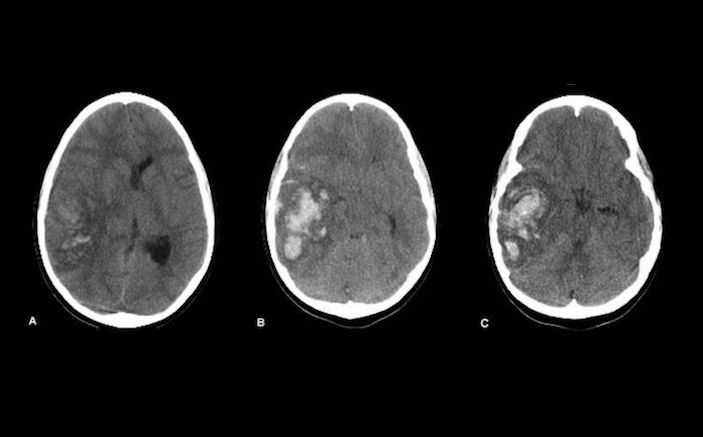
A 67-year-old woman is brought in by EMS after she developed a severe headache at home with associated vomiting and altered mental status. Her past medical history is significant for hypertension and atrial fibrillation, for which she takes atenolol and dabigatran (Pradaxa). The patient is somnolent, and after quickly securing her airway, you send her for a stat head CT. The CT shows an intraparenchymal hemorrhage. After paging your neurosurgeon on call, you consider your options for reversal of anticoagulation. You know you need to reverse her anticoagulation, but how?
The new oral anticoagulants (NOACs) are skyrocketing in popularity, displacing warfarin as first-line anticoagulants for many patients. Dabigatran is a direct thrombin inhibitor used for stroke prevention in patients with atrial fibrillation and for treatment and secondary prevention of venous thromboembolism. Dabigatran and other NOACs have several advantages over warfarin. Their pharmacokinetics are simpler, they don’t have the pesky food and drug interactions that plague warfarin, they don’t require monitoring and frequent dose adjustments, and importantly, they carry a lower risk of major bleeding complications.
However, a major concern is that unlike warfarin, which can be reversed with Prothrombin Complex Concentrates (PCC) or fresh frozen plasma and vitamin K, dabigatran and the other NOACs lacked a dedicated reversal agent. This made emergency physicians and others who anticipate worst case scenarios somewhat nervous. Although the NOACs are associated with fewer serious bleeding complications than warfarin, life-threatening bleeding can still occur. How should we manage the patient on dabigatran with an intracranial hemorrhage? Or the trauma patient who requires emergent surgery for their liver laceration who happens to be on dabigatran? Alternative reversal methods have been proposed, such as use of 3- or 4-factor PCC, fresh frozen plasma, or even emergent hemodialysis to remove circulating anticoagulant.However, the efficacy of these methods has been questioned, and safety concerns have been raised regarding thrombotic risk of PCC.
In October 2015 the U.S FDA approved a target-specific reversal agent for Dabigatran. Idarucizumab, marketed as Praxbind, is a monoclonal antibody fragment (Fab) that binds directly to dabigatran, neutralizing its activity. It is approved for reversal of anticoagulation in patients on dabigatran requiring emergent or urgent surgery or in patients with life-threatening bleeding. Dabigatran inhibits thrombin, which catalyzes one of the final steps in the clotting cascade. Idarucizumab reverses dabigatran’s anticoagulant effects by binding tightly to dabigatran with an affinity 350 times greater than thrombin, thus freeing thrombin’s functionality in the clotting cascade.
Studies show that administration of idarucizumab to healthy young volunteers, older volunteers aged 65-80, and volunteers aged 45-80 with mild or moderate renal impairment resulted in complete reversal of dabigatran’s anticoagulant effects within minutes without any procoagulant effects. This reversal of anticoagulation lasts 24 hours, which is an advantage over PCC, which has more transient effects. One important caveat is that there must be dabigatran in the bloodstream for idarucizumab to have any effect. Once the dabigatran is cleared by the kidney, idarucizumab will have nothing to bind to and will have no effect. It is recommended to give it if the last dose of dabigatran was in the last 24-48 hours. There may be some benefit of longer time frames in patients with renal failure, who will have a slower clearance rate of the dabigatran. Finally, since it is a monoclonal antibody, it is highly specific for dabigatran. It will not reverse the anticoagulant effects of coumadin, plavix, or other NOACs such as rivaroxaban.
The data on idarucizumab in patients who are actually bleeding or being operated on looks favorable as well. The Reversal Effects of Idarucizumab on Active Dabigaran (RE-VERSE AD) trial, a large international prospective cohort study of patients on dabigatran who receive idarucizumab either for serious bleeding or prior to an urgent surgical procedure, is still ongoing. A preliminary analysis of the first 90 patients revealed that idarucizumab rapidly and completely restored coagulation parameters in 88 to 98% of patients who had elevated clotting times at baseline. Among patients who underwent surgery, normal hemostasis was reported in 92%, with mild to moderate impairment in 8%. Only 1 of 90 patients (1%) had a thrombotic event within 72 hours of administration of idarucizumab. These data mirror the safety and efficacy data on idarucizumab from earlier human and animal studies.
Dosing
The FDA-approved dose is 5 mg, which is administered as two separate 2.5 mg IV doses infused over 5 minutes. The second dose should be administered within 15 minutes of the first infusion. There is no dosing change needed for renal or hepatic impairment.
Adverse Events
Adverse reactions are rare and include headache (5%) and hypokalemia (7%). There are case reports of serious complications in patients receiving idarucizumab, including acute ischemic stroke, cardiac arrest, NSTEMI, DVT, PE, but the incidence is thought to be extremely low.
Cautions
Contraindications include hypersensitivity to idarucizumab or any components of the formulation. Risks/benefits of anticoagulation should be considered before reversing anticoagulation with idarucizumab, since the underlying disease state may predispose to thrombotic events. However, since it is typically reserved for life-threatening bleeds, the scale typically would favor its use. Cost is also a consideration. Not all emergency departments may be able to stock it due to the infrequent need for it and its cost.
Pregnancy
There are no studies of human or animal models of pregnancy. It is unknown if idarucizumab is excreted in breast milk.
Cost
A single 2.5 mg/50 mL dose costs $2,100, so recommended treatment with two doses costs $4,200. This is slightly cheaper than 4-factor PCC, which costs about $5,000 for an 80 kg patient.
Disclaimer
The authors of this column receive no funding or incentives from any pharmaceutical company, and have no conflicts of interest related to the topics of their articles. Furthermore, Praxbind is not an advertising client of Emergency Physicians Monthly.
REFERENCES
- 1. Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;2009(361):1139-1151.
- 2. Alikhan R, Rayment R, Keeling D, et al. The acute management of haemorrhage, surgery and overdose in patients receiving dabigatran. Emerg Med J. 2014;31(2):163-168. doi: 10.1136/emermed-2012-201976 [doi].
- 3. Honickel M, Braunschweig T, van Ryn J, et al. Prothrombin complex concentrate is effective in treating the anticoagulant effects of dabigatran in a porcine polytrauma model. The Journal of the American Society of Anesthesiologists. 2015;123(6):1350-1361.
- 4. Singh T, Maw TT, Henry BL, et al. Extracorporeal therapy for dabigatran removal in the treatment of acute bleeding: A single center experience. Clin J Am Soc Nephrol. 2013;8(9):1533-1539. doi: 10.2215/CJN.01570213 [doi].
- 5. Honickel M, Treutler S, van Ryn J, Tillmann S, Rossaint R, Grottke O. Reversal of dabigatran anticoagulation ex vivo: Porcine study comparing prothrombin complex concentrates and idarucizumab. Thromb Haemost. 2015;113(4):728-740.
- 6. Lexicomp. idarucizumab: Drug information. www.uptodate.com Web site. 2017.
- 7. Pollack Jr CV, Reilly PA, Eikelboom J, et al. Idarucizumab for dabigatran reversal. N Engl J Med. 2015;373(6):511-520.
- 8. Gottlieb M, Khishfe B. Idarucizumab for the reversal of dabigatran. Ann Emerg Med. . doi: http://dx.doi.org.libproxy.lib.unc.edu/10.1016/j.annemergmed.2016.11.025.
- 9. Lexicomp. prothrombin complex concentrate, 4-factor, unactivated, from human plasma. www.uptodate.com Web site. 2017.