 In February of 2013, the American College of Emergency Physicians published a Clinical Policy statement regarding the use of tissue plasminogen activator (tPA, Alteplase®, Genentech) for the treatment of acute ischemic stroke.
In February of 2013, the American College of Emergency Physicians published a Clinical Policy statement regarding the use of tissue plasminogen activator (tPA, Alteplase®, Genentech) for the treatment of acute ischemic stroke.

In February of 2013, the American College of Emergency Physicians published a Clinical Policy statement regarding the use of tissue plasminogen activator (tPA, Alteplase®, Genentech) for the treatment of acute ischemic stroke. These guidelines – with a Level A recommendation for offering tPA within the 3-hour window following the National Institute of Neurologic Disorders and Stroke (NINDS) criteria, and a Level B recommendation within 4.5 hours – finally brought ACEP into line with guidelines established by the American Heart Association and the American Stroke Association.
However, months later, at the ACEP Scientific Assembly, the ACEP Council voted to “reconsider” this Clinical Policy and open the published statement for public comment. How did these guidelines elicit such rancor? Why, after decades of evolving evidence, does controversy in the treatment of acute ischemic stroke persist?
As one would expect, with over 18 years since the publication of the original NINDS trial, the issues of contention are well-established. The debate has settled into a persistent stalemate, with both viewpoints secure in the belief they are advocating in the best interests of patients. In a nutshell, the familiar claims put forth by tPA advocate and skeptics are listed in Table 1.

Of course, a Clinical Policy statement is not designed to accommodate contentious head-to-head debate. Rather, it serves as a tool for synthesizing the evidence into a concise format to define the standard of care. However, this clinical policy statement dispenses with a balanced view of the scientific debate in lieu of a prescriptive directive endorsing the use of tPA – both in the 3-hour window described in NINDS, as well as a 4.5-hour window not approved by the FDA. For a therapy predicated on a rush to evaluation and treatment, depriving emergency physicians of flexibility and medicolegal protection in the challenging diagnostic environment of acute neurologic emergencies carries substantial gravity. To do so, serious consideration must be taken as to the validity and integrity of the strong Level A and Level B recommendations described in this clinical policy.
Unfortunately, this policy statement is fundamentally flawed in its capacity to provide critical appraisal of the evidence relevant to the recommendations. In 2011, the Institute of Medicine published standards for the authorship of clinical guidelines. Entitled “Standards for Developing Trustworthy Clinical Practice Guidelines”, the core of this document aims to maximize transparency, eliminate conflicts-of-interest, and ensure adequate external review. The ACEP tPA policy, unfortunately, fails to meet many of the minimum standards for trustworthiness. The authorship panel is composed of several physicians with direct and indirect, financial and professional, conflicts-of-interest. Methodologic experts and patient advocates are under-represented, as is any point of view promoting cautious consideration of the risks of tPA. Many physicians are concerned that the composition of the authorship panel essentially pre-ordained the recommendations, rather than reflecting the balance of disagreement voiced over the last two decades.
 The strength of the recommendations also represents a significant issue to many emergency physicians. The Level A recommendation – “generally accepted principles for patient management that reflect a high degree of clinical certainty” – seems to suggest the debate has conclusively closed. As the presented and published opinions of many respected academic EPs will attest – including Jerome Hoffman, David Newman, David Schriger, and Anand Swaminathan – the evidence reflects anything but a “high degree of clinical certainty” favoring tPA. Looking beyond the borders of our North American island, we find last year the BMJ publishing a “Head to Head” debating the risks of thrombolytics in stroke, and the Australian and New Zealand professional societies stating the evidence in support of tPA remains insufficient. Clearly, the safe and ideal use of tPA remains a subject of international uncertainty, and a policy ought not unwaveringly dictate care in the presence of such ambiguity.
The strength of the recommendations also represents a significant issue to many emergency physicians. The Level A recommendation – “generally accepted principles for patient management that reflect a high degree of clinical certainty” – seems to suggest the debate has conclusively closed. As the presented and published opinions of many respected academic EPs will attest – including Jerome Hoffman, David Newman, David Schriger, and Anand Swaminathan – the evidence reflects anything but a “high degree of clinical certainty” favoring tPA. Looking beyond the borders of our North American island, we find last year the BMJ publishing a “Head to Head” debating the risks of thrombolytics in stroke, and the Australian and New Zealand professional societies stating the evidence in support of tPA remains insufficient. Clearly, the safe and ideal use of tPA remains a subject of international uncertainty, and a policy ought not unwaveringly dictate care in the presence of such ambiguity.
Furthermore, as any EP knows, patients presenting with acute ischemic stroke are widely heterogeneous – and the proposed likelihood of benefit and the risk of tPA are equally so. Advancing age, history of smoking, uncontrolled diabetes, prior disability, renal insufficiency and specific stroke syndromes all carry with them differing likelihood of sustained rapid reperfusion with tPA – as well as clinical worsening or death due to hemorrhagic transformation. There are no validated, endorsed tools available to assist EPs in assessing the individual risk and benefit for each patient. In a setting capable of providing rapid evaluation by a stroke neurologist, advanced imaging confirming vascular obstruction, and comprehensive institutional support, tPA may potentially be deployed in a judicious fashion maximizing its efficacy. In the absence of these resources, ACEP does not serve the needs of EPs by recommending this therapy without tools to assist in tweezing out the “indicated” from the “eligible”, and to support a shared decision-making conversation with patients and families.
Clinical policy statements and practice guidelines are powerful tools to reduce system variability. When a treatment is fully described – such as the thrombolytics trials for myocardial infarction enrolling nearly 20 times as many patients – the acceptability of guidelines is greatly enhanced. This ACEP Clinical Policy statement, however, falls dramatically short in many critical aspects necessary to guide the safe use of tPA. Until concerns regarding trustworthiness and scientific validity are fully addressed, our specialty, and our patients, will suffer.
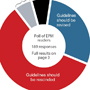
The public comment period for ACEP’s tPA guidelines began January 25th and will run for 60 days. All practicing emergency physicians should visit www.acep.org/commentform/IVtPA-Stroke to read and consider the published statement. Together, along with experts in emergency medicine, vascular neurology, and critical appraisal, we can work to revise this policy to better reflect the uncertainty surrounding safe and effective use of tPA. The ACEP Council took an unprecedented step in overwhelmingly rejecting this policy statement – and it is now the responsibility of each practicing EP to follow-up to protect the ability to use their best clinical judgment in their efforts to protect and care for patients.
Ryan P. Radecki, MD, MS, is an Assistant Professor of Emergency Medicine at the University of Texas Medical School at Houston. He blogs at emlitofnote.com and can be found on Twitter @emlitofnote. He has no professional or financial conflicts of interest to disclose.




7 Comments
Well said. This issue should not be laid to rest by a few, potentially biased commentators. Unfortunately, this provides ammunition to the legal community as well, who will skewer physicians who are trying to do the best possible for their patients.
Thank gawd someone in authority is addressing this insanity. We phone a big city neurologist, present our rural Iowa case, and are told, “Well, it’s up to you, Doctor.” My understanding is that it may save six, and kill six. Or some variation thereof.
A key factor in my skepticism of tPA is the distinct possibility that the patient before me, with focal deficits, may well improve on their own within hours, without tPA. I have observed this many times. Presumably such patients had TIA, complex migraine, etc. (I have seen 2 cases of a global insult presenting as focal deficits, due to carbon monoxide poisoning.) Had any of these people been given tPA, they would have been tPA “saves” (or perhaps would have had bleeds secondary to the medication).
I work at a Hospital that is a Stroke Center, and will only give TPA when backed into a corner with absolutely no contraindications. I have had a patient die after being in a vegetative state for 3 months caused by a TPA induced hemorrhagic stroke. Most of the time when I present this as a very risky and dangerous drug to patients and families, they will opt to take a safer route of therapy.
I agree that the rush to standardize TPA for stroke is premature. I am bothered by the lack of confirmatory studies, and by the low therapeutic index of this therapy. I personally feel that we need more tools to better define who will benefit from TPA vs. who will not (and be unnecessarily exposed to the risk of hemorrhagic transformation).
I work in a community hospital with 3 neurologist and one of them active and we see about 600 strokes in a year. Our tPA rate is around below 10%.
The concerns I have is the guidelines to slect the patients for thrombolyitcs should be a team approch.
Most of the times the decision is left to the ED physician and neurologist are available on the phone.
Patient care does not end with thrombolytics, there should be continuation of treatment after ED, this lacking in 90% of the community hospitals.
Patient should be transferred to a multidisplinary institutions for further care.
Excellent! Stay on it til the right study is done w/ NO COI and primarily proctored/set up by METHODOLOGISTS!
This issue is not dead.
Where do these neurologists come from? What science to they use?? Are any of them unbiased?
Thanks!