A new innovation in brain injury evaluation using biomarkers.
***This article was written by one of EPM’s advertising partners as a promotional supplement.***
We don’t often use words like “breakthrough” or “game changer,” but when we do — and this is both, you don’t want to miss it. So here it is in a nutshell.
Abbott has developed a blood plasma test that picks up biomarkers of traumatic brain injury. For decades our ability to detect traumatic brain injury was limited to indirect measures of function such as the Glasgow Coma Scale, history of loss of consciousness, impaired memory, changes in mentation, etc. Our main concern was whether a CT scan was indicated to detect a neurosurgical emergency. That’s going to change.

Abbott’s i-STAT TBI Plasma test uses the next generation i-STAT Alinity and measures the biomarkers GFAP (glial fibrillary acidic protein) and UCH-L1 (ubiquitin carboxy-
terminal hydrolase L1). GFAP is a protein expressed by numerous cell types in the central nervous system, including astrocytes and ependymal cells. Elevation of GFAP
occurs in astroglilial injuries and is specific to the central nervous system.[1]
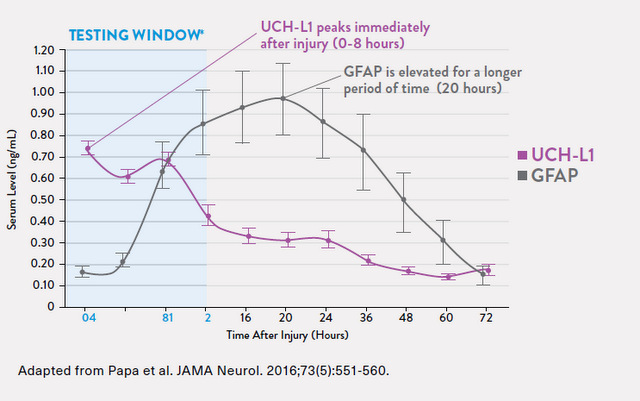
There are other biomarkers detectable in brain injury. But these two, like the currently used biomarkers in heart muscle injury, rise and fall in curves that will detect brain injuries in the hours after injury. The test is intended for use within 12 hours after injury; UCH-L1 rises immediately after injury and falls after several hours while GFAP take longer to rise and stays elevated for a longer period of time.[2]
How important is a test of this nature? An estimated 4.8 million emergency room visits per year present with the complaint of head injury, concussion, sports injuries, and slip and-falls.[3] The question of loss of consciousness is seldom answered reliably and the GCS score is very insensitive for all but major injuries.
The value of the i-STAT TBI Plasma test is seen as a way to speed potential concussion patients through the ED. Test results are available within 15 minutes after the plasma
is placed in a test cartridge. Over the last year of the pandemic, this was seen as a particular advantage, getting elderly slip-and-fall patients out of the ED quickly and possibly avoiding the unnecessary radiation and potential infectious exposure.
Research demonstrated that many CTs could be avoided. In fact the pivotal studies suggest a potential reduction in unnecessary CT scans of up to 40%. [4] While the i-STAT Alinity handheld analyzer is portable and the test takes only 15 minutes, it still
requires that blood specimens be spun down to plasma, requiring the process to take place in a laboratory.

It is anticipated that a whole blood test could be available in the future that would be portable and usable for head injuries requiring quick evaluation, such as sports
injuries. As would be expected, the military has been interested in the development of such a test for over a decade and played a critical role in the development of Abbott’s
i-STAT TBI Plasma test.
The Transforming Research and Clinical Knowledge in Traumatic Brain Injury (TRACK-TBI) study, is one of the largest TBI studies of its kind. Geoffrey T. Manley MD, PhD, the principal investigator in the TRACK-TBI study, is a professor of neurosurgery at the University of California, San Francisco.
“Blood based biomarkers are emerging as an important tool to detect TBI, and this research opens the next chapter for how this condition is evaluated,” said Dr. Manley.
“Having these sensitive tools could provide physicians more real-time, objective information and improve the accuracy of detecting TBI,” Manley said. “This research
shows that blood tests have the potential to help physicians triage patients suspected of brain injury quickly and accurately.”
Here’s the bottom line. Just as cardiac biomarkers changed how we practice in emergency cardiac care, brain biomarkers are likely to change how we treat traumatic brain injury. This is a game changer.
References:
1. Metting et al. Neurology. 2012;78(18):1428-1433.
2. Papa L et al. JAMA Neurol. 2016;73(5):551-560.
3. Korley FK et al. J Head Trauma Rehabil. 2016;31(6):379-387.
4. i-STAT TBI Plasma Cartridge. Instructions for use. Abbott Point of Care Inc. Abbott Park, IL; 2021.