Keep an eye or two on the Diaton Tonometer.
**The following is a paid promotional piece from one of EPM’s sponsors.**
Measuring the intraocular pressure (IOP) no longer requires placing something on the surface of the cornea. The FDA has approved the use of a new device, called Diaton Tonometer, which measures IOP through the eyelid and sclera. Don’t worry about placing anesthetic drops in the eye, which present a risk of accidental post measurement injury to the cornea. The Diaton Tonometer also removes the need for expensive replacement of one-time-use tonometer covers. Nor will you be bothered with time consuming recalibration of the tonometer before each new patient.
With millions of patients presenting to the ED that have had some type of corneal refractive surgery, such as LASIK or RK, which soften the cornea and distorts the IOP readings; transpalpebral and scleral measurements of IOP may actually be more accurate.
Game Changer
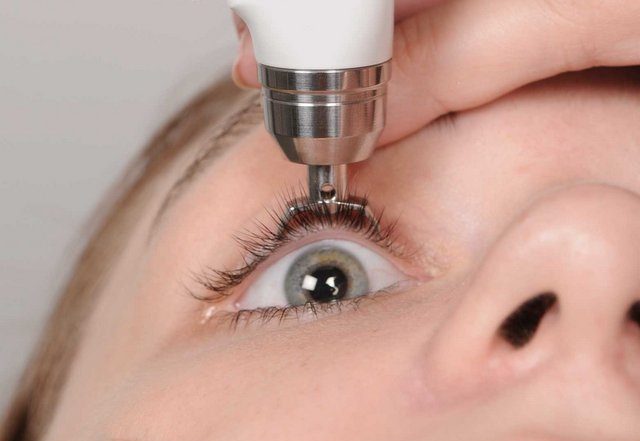
Transpalpebral measurement of IOP is a game changer in a field that has seen very little major innovation since the invention of the Schiotz tonometer decades ago. Diaton is a pen-like device with a stainless steel tip that simply touches the lid in the area above the corneal margin, at the sclera. The patient can be in a chair or gurney. A couple of confirmatory measurements and you’re done. Diaton is inspired by Dr. Svyatoslav Fyodorov, a Russian ophthalmologist, who is credited with inventing radial keratotomy (RK) procedure that became widely used in the US. Transpalpebral tonometry similarly changes the availability to diagnose and monitor a host of intraocular pathologies.
In the emergency department, acute angle-closure glaucoma should be considered in the differential diagnosis with any patient presenting with eye pain, headache, blurred vision, visual haloes, nausea, vomiting or even abdominal pain. Failing to measure the IOP is to risk a failure to diagnose a condition that result in blindness, let alone a major liability suit. But a simple IOP measurement is often overlooked because of the logistical hurdles.

If you’ve ever tried to measure the IOP of someone, like a child, who steadfastly refuses to fully open their eye, due to pain, fear, or injury, you understand the challenge of something as simple as instilling eye drops. Then you have to worry about the secondary effects of the drops, desiccation of the lens, inadvertent corneal abrasion and complications due to anesthesia of the cornea. Now you don’t have to. You can reassure the patient that they will not have anything in their eye, which greatly reduces patient hesitation. The operator technique for using the Diaton can be learned in a few minutes and mastered very quickly.
An interesting history
Ukrainian native Roman Iospa, who immigrated to the US as a teenager, brought the Diaton tonometer to the American market. As a native Russian speaker with an education in finance during the Dotcom boom, Roman interfaced with many Russian entrepreneurs seeking to open the American market to Russian products and services. The Diaton tonometer caught Roman’s attention when a friend was diagnosed with acute narrow angle glaucoma that required constant monitoring and management. That’s when he became aware of the importance of the ability to measure IOP quickly, easily and without any eye drops, risk of infection or contact with the cornea. Roman became the head of operations and eventually CEO in charge of the US and international market development.
The Diaton tonometer was first introduced to ophthalmologists and optometrists before approaching the emergency medicine market. But, as in all new technologies, they have faced challenges in changing what is seen as ‘standard practice.’

I was first introduced to the Diaton tonometer at the welcome mixer at the 2019 ACEP Scientific Assembly in Denver and was fascinated by Roman’s personal story. But when he told me about transpalpebral tonometry, I have to admit, I didn’t believe him. So right there, he took out the device and demonstrated it on me, with no anesthesia — except maybe my first beer — how easily the device could be used. I was amazed.
Does it Work?
The Diaton tonometer was introduced to the American market after extensive clinical studies and clearance from the FDA. Research presented by Dr. Mark Latina, a glaucoma specialist at Harvard’s Massachusetts Eye & Ear Infirmary found the Diaton tonometer provided similar results to those made using Goldmann applanation, the “gold standard of tonometry,” but without the operator challenges of that method.
One of the added perks of the transpalpebral method is that it does not require the patient to remove contact lenses, as the measurement is taken at the sclera, just above the lens.
New applications and settings
The ease of transpalpebral measurement of IOP opens the door to some new and significantly wider applications. Without concern for transfer of eye infections, transpalpebral measurement of IOP can now be used to easily screen thousands of patients in the US as well as developing countries.

Are you an Early Adopter?
It’s a simple change in technology that solves a variety of very old problems. I suspect the Diaton tonometer will become the standard of care for measurement of IOP in EDs eventually. The only question is how fast EPs will recognize its utility and improvement over the current methods and take the steps to incorporate it into their daily practices.
To obtain more information about the Diaton tonometer:
Visit: www.TonometerDiaton.com
Phone: 877-342-8667
Email: contact@tonometerdiaton.com

1 Comment
A Big thanks to EP Monthly for Diaton tonometer coverage.
The Diaton Raises Standard of Care with its Transpalpebral Scleral Tonometry technology.
This breakthrough innovation allows to accurately measure intra-ocular pressure (IOP) independent of CCT and other corneal biomechanics.
Diaton tonometer meets and exceeds Joint Commission Guideline for Disinfection and Sterilization Guidelines and Standards as Diaton has no contact with an eyeball or the mucous membrane.
The Joint Commission, Division of Healthcare Improvement in Issue 49, May 2019 noted that “Tonometer tips are particularly problematic because disinfectants can dissolve the glue that holds the hollow tip together, causing the tip to swell and crack. It’s important to note that tonometer tips have been identified as sources of ophthalmic nosocomial outbreaks commonly linked to adenovirus types 8 and 19. Desiccated virus remains viable and can be recovered after 49 days on dried plastic or metal surfaces.
Areas that can Benefit from Sterile Diaton tonometer:
Emergency departments
Urgent care centers
Ophthalmology clinics, optometrist offices, and procedure rooms
Neonatal intensive care units (NICUs)
“Items that touch mucous membranes — such as the eye — must be, at minimum, high-level disinfected. Items that contact or enter sterile tissues — such as instruments that are used for surgical procedures — or touch an ulcerated cornea must be sterilized. ”
The Joint Commission, Division of Healthcare Improvement
FDA cleared Scleral and Transpalpebral tonometry technology shows indisputable advantages vs All other contact and corneal tonometers which require direct contact with the cornea. Since there is No Contact with an eyeball, Risk of Infection or Cross Contamination is Null with Diaton’s technology.
Another Key Item is being overlooked in ED ER Setting is that variability in Corneal Biomechanics influences All other corneal or contact devices, but not the Diaton:
Diaton measures Accurate IOP in Eyes that are affected by corneal pathologies which make corneal tonometry inaccurate, and often impossible – corneal injury, corneal edema, keratoconus, atypical corneal thickness, corneal rigidity, keratitis, cornea dimness, post keratoplastics, keratoprosthesis, post LASIK, high degree of ametropy, astigmatism and many other conditions which walk into Emergency Departments and Emergency Rooms.
Diaton tonometer can measure accurate IOP with contact or scleral lenses still being on the eye.
The American Academy of Ophthalmology has reported that transmission of adenovirus and herpes simplex virus HIV, hepatitis C virus (HCV), enterovirus 70, Pseudomonas aeruginosa, methicillin-resistant Staphylococcus aureus, Acanthamoeba, and prions (transmissible spongiform encephalopathies, such as Creutzfeldt-Jakob disease) could occur from failure to adequately disinfect ophthalmology devices, such as tonometers.
Infection Control Departments in many hospitals have been satisfied with an affective way Diaton tonometer compliance for infection control.
Learn more at https://diaton.com