When a patient presenting a fever has traveled abroad recently, your diagnostician’s “spidey sense” should be tingling wildly. Here’s a guide to the potential disease culprits, current issues and how to respond.
The incredible ease with which people travel across the world today means that communicable diseases spread just as easily, far more so than in the past. Some of these diseases – among them malaria, dengue and MERS-CoV – can be life-threatening. Considering this, the emergency physician must understand how to assess a returning traveler who has a fever. Not only is it important to understand disease symptomology and how to treat your patients, but it’s essential to know how to protect your staff and other patients from becoming infected. Universal precautions should always be followed; however, some of these diseases require heightened levels of isolation.
When assessing returning travelers, a few key questions will help guide your evaluation and management of the patient: Where did they travel to? Which pre-departure vaccines did they receive? Did they take malaria prophylaxis and did they take it properly? Finally, how long has it been since they returned?
Several websites and online sources can help guide your assessment of the returning traveler with a fever. Important resources include the CDC and the CDC Yellow Book [1]. Another helpful site is Healthmap. This site lists most of the ongoing epidemics worldwide. If you know the area your patient visited, you can look up likely exposures. If you’re interested in tracking emerging diseases, ProMED Digest is another great source of information. Often new diseases and new epidemics are initially reported on ProMED.
Knowing if your patient received pre-departure vaccines helps eliminate, or at least lessen, the likelihood of several diseases. The yellow fever vaccine is very effective, and patients who receive it at least 10 days prior to travel are unlikely to contract yellow fever. Other potential pre-departure vaccines include typhoid, meningococcal, rabies, tetanus and Japanese encephalitis. Pre-departure vaccine recommendations are based on where the patient is planning to travel. Patients should also be up to date on their general vaccinations including tetanus and the flu vaccine.
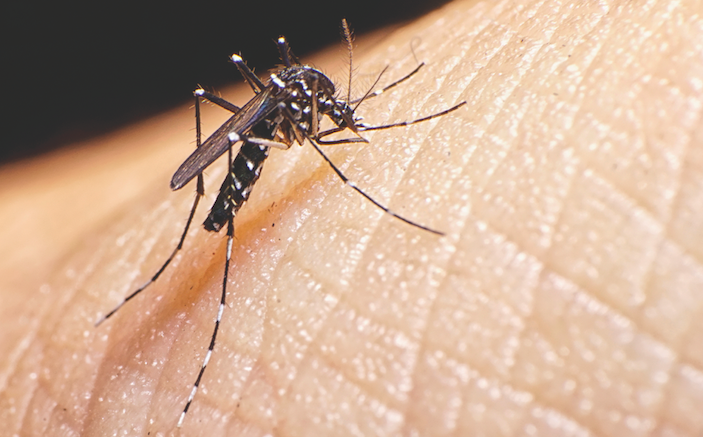
There are several diseases that we should consider when diagnosing returning travelers. First and foremost, common illnesses are, well, common. Most of these patients will not have a tropical disease, but will have typical viral illnesses, influenza or exacerbations of their chronic diseases. Other worldwide diseases that should be considered include malaria, dengue and chikungunya. Middle East Respiratory Syndrome-Coronavirus (MERS-CoV) and avian influenza are also a concern. Zika virus has been spreading north in the Americas since April 2015. In January, a case was reported in Texas, and, in the same month, physicians found that a Hawaii baby born with microcephaly had been infected with the disease.
Dengue, chikungunya, Zika and malaria all present with very similar symptoms. There are serologic tests available that can confirm your diagnosis but most are send-out labs and treatment is time-sensitive. For instance, if there is any question that the patient may have malaria, he should be started on antimalarial treatment pending laboratory confirmation. Patients should also be assessed for neutropenia, thrombocytopenia, anemia, coagulopathies and electrolyte abnormalities.
Given this variety of diseases, some familiar and some foreign, the fundamental question becomes: How do we recognize these patients, assess them and treat them? An accurate diagnosis starts with having a high index of suspicion; if you do not suspect these disease processes, you won’t identify them in your patients.
 Malaria
Malaria
Malaria is one of the more common tropical diseases seen in returning travelers. It can be caused by six known species of Plasmodium and is spread by the Anopheles mosquito. This mosquito bites at night; thus, people who sleep in screened or air-conditioned rooms have a lower risk of contracting the disease. Malaria has a typical incubation period of 8-25 days; one species, P. vivax, can have an incubation period of up to 12 months. The most likely species of malaria causing disease in your patient is dependent on the region he visited (see guide to strains below).
There are several different pre-departure regimens of malaria chemoprophylaxis that may have been prescribed to your patient. Selection of a particular regimen depends on your patient, his age and the area he visited. The CDC lists some of the more common chemoprophylaxes. Asking your patient what he took, when he took it, if he took the entire course of therapy and where he obtained the medicine will help determine your patient’s risk. Most regimens require the traveler to take medications before, during and after travel. Patients who were prescribed inappropriate chemoprophylaxis or who do not complete the entire course of therapy are at increased risk of infection. As counterfeit antimalarial medicines are a significant problem in certain countries, knowing if your patient purchased his medicine overseas is essential. None of the regimens are 100% successful in preventing malaria even if taken correctly.
Patients will usually present to the ED with headaches, fever and joint pain. They may have jaundice and evidence of hemolytic anemia. The fever may be the classic cyclic fever described with malaria that recurs every 2-3 days, or it may be continuous. The disease itself can be categorized as uncomplicated or severe. Severe malaria is characterized by significant organ dysfunction, anemia, electrolyte abnormalities or cerebral malaria (altered mental status, seizures or coma). Severe malaria is a medical emergency that must be treated aggressively.
Diagnosis is made by thick and thin smears, which take time to prepare and read. Therefore, if malaria is suspected, especially severe malaria, empiric treatment should be initiated immediately while awaiting the results of the blood smears. There are also rapid antigen tests available to diagnose malaria within 20 minutes. However, since these tests have a significant number of false positives, they should be confirmed by blood smears. Other abnormalities evidenced in the patient include hemolytic anemia, thrombocytopenia, hepatic injury and renal failure; thus, a CBC, liver function panel, a basic metabolic panel and a urinalysis looking for hematuria may help guide you to your diagnosis.
Treatment of malaria is dependent on the strain and the region the patient visited (see sidebar describing the six known strains). The CDC has a nice algorithm to help you manage malaria. For uncomplicated malaria, the patient can usually be treated with oral medications. In general, any patient with P. falciparum, knowlesi or an undifferentiated strain will require admission to monitor disease progression. In contrast, stable patients known to have P. malariae, ovale or vivax can be managed as outpatients. For falciparum, the oral management includes artemisinin in combination with 1-2 other medications such as lumefantrine, mefloquine, amodiaquine or sulfadoxine/pyrimethamine. The remaining strains can be managed with chloroquine. There is currently a nationwide shortage of chloroquine, so it’s important to consider alternatives. Hydroxychloroquine, a drug commonly used to treat rheumatoid arthritis, might be easier to obtain in US pharmacies and can be substituted for chloroquine. To prevent relapses, primaquine should be added to eradicate the dormant liver form of the parasite for P. vivax or ovale infections. In patients with G6PD deficiency, primaquine can incite hemolysis, so patients should be tested for G6PD deficiency before being treated with primaquine. G6PD testing can often be performed in larger facilities but is a send-out lab in many smaller hospitals. There is a rapid qualitative test for G6PD that is accurate in malarial endemic areas but is not typically available in U.S. hospitals.
Patients with severe malaria will need ICU admission and IV antimalarial treatment. They often need blood transfusions as well. Worldwide, the most commonly used IV antimalarial is artesunate; however, in the United States, this drug is only available through the CDC. Treatment can be initiated with quinidine plus doxycycline or clindamycin IV. IV quinidine is cardiotoxic and can cause hypotension, ventricular arrhythmias and QT prolongation; thus, the patient should be on a cardiac monitor during administration. If quinidine is not available, the CDC should be contacted to obtain artesunate.
Malaria is usually treatable, but severe malaria can have a mortality of up to 20% when treated, especially if associated with cerebral malaria. The most important factors in decreasing mortality are recognition of the disease, rapid treatment initiation and close intensive care monitoring.
 Disease Culprits: A Guide to Malaria Strains
Disease Culprits: A Guide to Malaria Strains
Six known species of Plasmodium cause malaria. P. falciparum, the most lethal of all the species, is present in South East Asia (SEA), Sub-Saharan Africa, the Caribbean, and South and Central America. Falciparum is chloroquine-resistant in all areas except in Central America west of the Panama Canal, Haiti, the Dominican Republic, and parts of the Middle East. Of more concern, multidrug resistant falciparum is emerging in areas of Cambodia and Myanmar. P. malariae has a lower morbidity and mortality and is endemic in the same areas as falciparum except Central America. P. vivax, P. ovale curtisi and P. ovale wallikeri also have lower mortalities, but can have significant morbidity due to relapsing disease caused by parasites stored in the liver. All three are found in SEA, Africa and South and Central America. P. knowlesi, found almost exclusively in SEA, is morphologically similar to P. malariae through a microscope but has a mortality similar to falciparum.
When treating a patient with P. knowlesi think, “looks like malariae; acts like falciparum.” Fortunately, with the exception of chloroquine-resistant P. vivax in Papua New Guinea and Indonesia, non-falciparum strains of malaria continue to be sensitive to chloroquine.
 Dengue
Dengue
Dengue, or breakbone fever, is a viral hemorrhagic fever and carries a high morbidity and mortality. The incubation period is 3-14 days, with most patients presenting within the first week. There are four different syndromes that can be caused by dengue: undifferentiated infection, dengue fever, dengue hemorrhagic fever and dengue septic shock. Each syndrome is progressively more severe. There are five known strains of dengue: DENV-1 through DENV-5. Infection with one strain confers immunity to that specific strain; however, it offers no protection from other strains. In fact, subsequent infection with a different strain often results in a more severe infection.
The mild form of dengue, undifferentiated fever, is a non-specific flu-like illness and is self-limited. Dengue fever presents with a high fever, headache, rash and severe joint pain. The treatment is supportive care and pain control. Patients usually recover without significant sequelae and do not require admission if the diagnosis is clear.
Dengue hemorrhagic fever (DHF) and dengue septic shock (DSS) are the most severe syndromes. The febrile phase of DHF lasts up to 7 days with symptoms of fever, nausea, vomiting, headache and joint pain. Patients may develop petechiae or the classic dengue rash described as “islands of white in a sea of red.” Treatment includes pain medications (NSAIDS are contraindicated), fluid hydration and blood transfusions if needed. Most patients progress through the febrile phase and recover without significant sequelae. Some evolve into dengue septic shock.
Dengue septic shock occurs as the patient defervesces; it lasts two to three days and has a high mortality and morbidity. Patients develop a massive plasma leak leading to pleural effusions, ascites and hypovolemic shock. Patients also develop DIC with massive hemorrhage. Treatment is with blood and judicious fluids. In both DHF and DSS, it is important to monitor renal function, hematocrit and platelets, as these patients will be thrombocytopenic. Patients who survive then progress to a convalescent phase lasting 2-3 days. During this stage, there is reabsorption of third-spaced fluid, which can lead to significant volume overload and result in cerebral edema. Therefore, IV hydration should be stopped when the patient transitions into the convalescent phase. Most patients who survive the convalescent period generally have few sequelae.
 Zika
Zika
Zika virus, spread by the mosquito Aedes aegypti, had never been seen in the Americas before it was detected in Brazil in April of 2015. It is believed that the virus was brought to Brazil by travelers coming to the 2014 FIFA World Cup. Prior to 2015, Zika had been primarily seen in Asia and Africa. Yet there have now been multiple outbreaks in several countries in South and Central America. As of the end of 2015, there have been cases as far north as Mexico, including the Yucatan and Baja peninsulas, as well as cases in Puerto Rico.
Zika has an incubation period of 2-14 days and presents with headache, fever, a maculopapular rash, malaise, arthralgias and conjunctivitis. Infection tends to be mild with low morbidity and mortality. However, there has been an increase in cases of Guillain-Barré syndrome following outbreaks of Zika in several countries. Also, since the Zika outbreak in Brazil, there has been a tenfold increase in the number of infants born with microcephaly. Similar CNS malformations were seen in neonates after a Zika outbreak in French Polynesia and other countries. Thus, our biggest concern with Zika may be its teratogenic effects and increased risk of developing GBS. Furthermore, unlike dengue or chikungunya, Zika can be transmitted human to human via sexual contact. Currently the management of Zika is simple supportive care with pain and fever control, and antihistamines as needed for itching. NSAIDS and ASA should be avoided due to potential for increased bleeding risk. It is also important to isolate these patients from pregnant women. The only human-to-human transmission to date has been via sexual contact, but given the TORCH effects of the virus, it is best not to expose pregnant staff to these patients. There has also been a concern for transmission of the virus via blood products, so universal precautions should always be followed when caring for these patients.
These are just some of the diseases you need to be aware of when diagnosing patients. Next month I’ll examine chikungunya, influenza-like illnesses and provide tips on vaccine prevention and patient assessment.
REFERENCE
- http://wwwnc.cdc.gov/travel/yellowbook/2016/post-travel-evaluation/fever-in-returned-travelers