During the most recent Scientific Assembly, IncubatED featured a virtual ‘shark tank’ competition highlighting four medical devices. These companies feature a portable MRI device, tools for securing IV access and draining abscesses, and point-of-care testing for coagulopathies. ACEP also launched their new EMergence podcast, which focuses on innovation for the Emergency Department.
Hyperfine’s Swoop™

Is there a way to get Magnetic Resonance Imaging (MRI) with the speed of point-of-care testing? The team at Hyperfine created Swoop to meet that challenge. Their Portable MR Imaging System is mobile, fits in elevators and through 34 inch doors, and can be moved right into a patient’s room.
It is powered with a standard electrical outlet, interfaces with an iPad, and provides initial neuroimages for the treatment team in minutes. Hyperfine’s goal is to make MRI machines portable and affordable. Swoop does not depend on a large budget, an imaging suite, patient transportation, or a dedicated staff.
In contrast to existing technology, Swoop was created to image critical patients in trauma rooms where they can continue to be monitored and treated. It is also able to capture those images despite the large amounts of metal in those rooms.
Patients can even hold hands with family members while being imaged. You can learn more at their website, see their ACEP20 pitch, and listen to the ACEP EMergence podcast with Dr. Khan Siddiqui, Radiologist and Chief Strategy Officer, and Dr. Charles Wira, Emergency Physician.
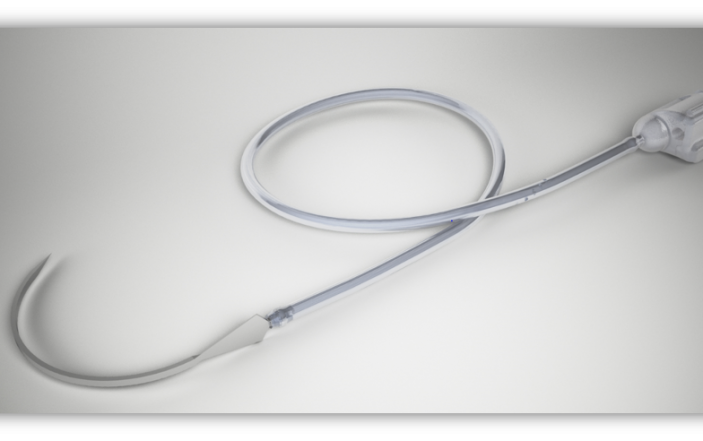
Lineus Medical’s SafeBreak® Vascular

SafeBreak Vascular was created to reduce IV catheter complications. When forces are applied to the IV line during patient movement or transport, SafeBreak intentionally separates to protect the IV catheter. Because IV access is used to draw labs, give fluids, and administer medication and blood products, this access is critical for patients who are a ‘tough stick.’
Access can be particularly tenuous for pediatric patients and for those with cognitive delays or altered mental status. SafeBreak is the only FDA-cleared Force-Activated Separation Device that reduces mechanical IV complications requiring new IV access. While 46% of peripheral IVs will fail, SafeBreak was able to reduce that by 44%. Their device prevents repeat needle sticks, saves time for nursing staff, and prevents medical supply waste. To learn more about SafeBreak, visit their website and check out their ACEP20 pitch.
EM Device Lab’s Quickloop

Quickloop was designed by Dr. Michael Gorn, a Pediatric Emergency Physician from Austin, TX, as an all-in-one tool that can be used to treat abscesses. This device is based on the clinically validated loop drainage technique, which a 2020 meta-analysis showed to have 42% decreased failure rate when compared to the traditional incision and drainage (I&D) technique.
The Quickloop all-in-one device incorporates an introducer needle, tubing with fenestrations for easy irrigation, and a hub to form a permanent loop and facilitate attachment to a syringe. This replaces the multi-component I&D kit, wound packing, and other tools needed for the conventional technique.
Using just one tool, the EM Device provides an easy way to increase treatment success, cut treatment time, reduce follow-up visits, cause less pain, and improve patient satisfaction. To learn more about Quickloop and EM Device Labs, listen to Dr. Gorn talk about the process of going from having an idea to launching a medical product on the Mastering Medical Device podcast, visit their website, and check out their ACEP20 pitch.
Coagulo
Coagulo pioneered lab testing for coagulopathies that is more affordable, gives results in the ED with point-of-care testing, and focuses on specific steps of the coagulation pathway. Traditional coagulopathy labwork focus on patients using coumadin and heparin, and give results based on Prothrombin Time (PT), International Normalized Ratio (INR), and Partial Thromboplastin Time (PTT).
As more patients are taking Direct Oral Anticoagulants (DOACs), Coagulo addresses this with results focused on the action of Factor IIa and Factor Xa. With Coagulo’s testing technology, drops of blood can placed one a single-use cartridge and results are available in 10 minutes.
All of this can be done in the ED without requiring complicated sample preparation or result calculations. Most importantly, by analyzing specific coagulation components, Coagulo testing provides information about coagulopathy due to medication, trauma, and sepsis. To learn more about Coagulo, visit their website, view their ACEP20 pitch, and listen to their ACEP Emergency podcast featuring their Founder, Dr. Galit Frydman.
References:
- Gottlieb, M., Schmitz, G., & Peksa, G. D. (2020). Comparison of the Loop Technique With Incision and Drainage for Skin and Soft Tissue Abscesses: A Systematic Review and Meta‐Academic Emergency Medicine, 28(3), 346–354. https://doi.org/10.1111/acem.14151