A potentially fatal dermatologic problem.
Case Report:
A three-year-old healthy female presented to the emergency department with the chief complaint of fever, sore throat and a diffuse rash for a period of five days. Her past medical history is not significant for any major illness and she is up-to-date with her immunizations. The patient was evaluated three days prior at her pediatrician’s office. A rapid strep screen was positive, and she was subsequently started on amoxicillin and was discharged.
Her first dose of antibiotics was that evening, and by the next day, about 12 hours later, she developed a rash on her cheeks and nose. The rash continued to get progressively worse to involve the trunk and torso with associated peeling. A review of systems was notable for a fever up to 101.6°F, sore throat, fatigue and a peeling rash.
Vitals on ED arrival were: temperature of 99.4°F (acetaminophen given prior to arrival), blood pressure of 80/53 mmHg, heart rate of 167 beats/min, respiratory rate of 50 breaths/min, weight of 13.6 kg, and a pulse oximetry of 100% on room air.
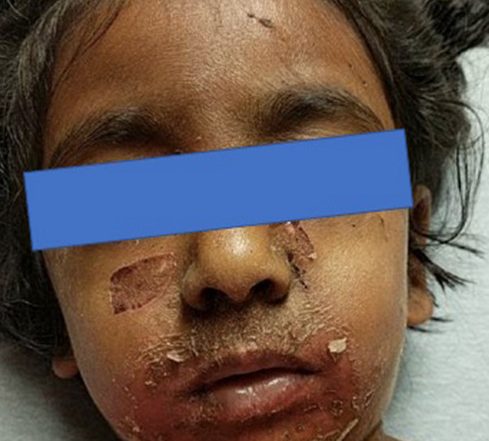
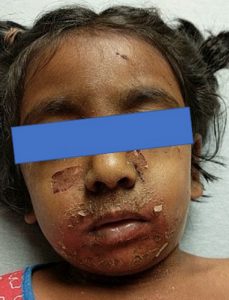
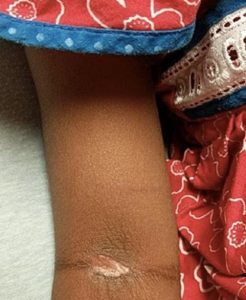
On physical exam, the patient appeared ill, crying and scared, but in no acute distress. She exhibited a diffuse erythematous rash on the arms, legs and torso with areas of desquamation and ruptured bullae involving the face (Image A), neck, anterior chest, axilla and antecubital fossa. The lesions were acutely tender to palpation with a positive Nikolsky’s sign (Image B).
Multiple areas of early erosion were noted with crusting on the lips. Conjunctivae were injected bilaterally with a small amount of drainage. Labs were notable for a leukocytosis of 14.4. A capillary blood gas revealed a pH of 7.41, pCO2 29 (35-45 mmHg), p02 75 (80-100), HCO3 18 (19-27 mmol/L), and a base excess of -5 (-2-2 mmol/L). Remaining laboratory evaluation proved unremarkable. The patient was started on 20 cc/kg of fluid resuscitation and blood cultures were obtained. The patient was ultimately transferred to the pediatric burn center.
The patient underwent continued intravenous fluid resuscitation, debridement, daily dressing changes, and daily monitoring for signs of infection and ocular involvement. By day 10, she continued to undergo emollients and massage with all sites rapidly healing. The patient and family were instructed on continued emollients and sun protection until all areas were blended in. At her two-month pediatric follow-up office visit, the patient has progressed well — skin lesions have completely resolved and she has returned to pre-school.
Discussion:
Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) are life-threatening dermatological emergencies characterized by widespread necrosis, detachment of the epidermis, mucous membrane erosion, and bullous skin lesions. The beta-lactam class of antibiotics, such as penicillin in this case, is known to cause SJS.
In the United States, the incidence of SJS between 2009 and 2012 has been reported at 5.3 cases per 1 million children every year.(1) In comparison, TEN has 0.4 cases per one million children every year.(2) In children and in adults, the foremost trigger of SJS/TEN is the use of medications. The most common offending drugs were antibiotics (29.5%) and anticonvulsants (24.1%). Carbamazepine, allopurinol, and penicillins are the most common single offending drugs (17.5%, 19.6 and 7.2%).(3) SJS/TEN affects any age, and it is more common in female patients.
SJS and TEN are considered a continuum of disease and identified based on the severity and the total percentage of body surface involved.(1) SJS is characterized by skin detachment involving <10% of the total body surface area (mucosal involvement in at least two locations: ocular, genital, oral). TEN is characterized by skin detachment involving >30% of the total body surface area. Between 10 percent to 30 percent, there is an area of overlap with both disease processes.4 In both of these cases, mucus membranes (ocular, genital, oral) are involved in up to 90% of all cases.(1)
The mechanism behind this potentially fatal dermatologic emergency is not fully understood. It is believed to be the result of an immune mediated hypersensitivity reaction involving cytotoxic T lymphocytes killing keratinocytes.(5,6)
In the ED, this is often a manifestation of drug-induced hypersensitivity. The average exposure period from starting the medication until the initial reaction has said to range from four days up to four weeks, with continuous use of the medication.(2) Medications that have been linked to SJS and TEN include, but not limited to:
- Sulfamethoxazole
- Anti-epileptics (carbamazepine, phenytoin, phenobarbital, lamotrigine)
- Diclofenac
- Tetracycline (doxycycline)
- Penicillin (amoxicillin/ampicillin)
- Fluoroquinolones (ciprofloxacin, levofloxacin)
- Rifampin
- There have been rare causes of SJS and TEN caused by Mycoplasma pneumoniae, cytomegalovirus and herpes virus.3,4 It should be noted that in over one-third of all cases, no identifiable cause could be found.
SJS Breakdown
After the initiating of antibiotics, SJS will present with a prodromal period consisting of fever, malaise, arthralgia and myalgia. Next, patients will begin to develop mucocutaneous lesions one to three days later. Some of the early subjective symptoms to aid the clinician for early mucosal involvement include conjunctival itching/burning, photophobia or odynophagia.(2,4,7) Mucosal involvement is seen in 90% of all cases of SJS or TEN. The oral mucosa and vermillion border are almost always involved.
Associated physical exam findings include hemorrhagic erosions, mucositis and stomatitis. Ocular involvement ranges from severe conjunctivitis, purulent discharge, corneal ulcerations, to suppurative keratitis and endophthalmitis.(2) Urogenital involvement can be seen in up to two-thirds of patients and consists of urethritis, urinary retention, ulcerative vaginitis and vulvar bullae, to name a few.(2) Involvement of the mucosa of the posterior pharynx is involved in nearly every patient.
Cutaneous involvement begins as erythematous macules with a purpuric center and diffuse erythema. Examination of the skin will reveal it to be exquisitely tender and pain will often be out of proportion to the physical exam findings.
As the disease process progresses, bullae begin to form, and the skin begins to slough off.(2) It is at this time that a patient may have a positive Nikolsky’s sign (extending an area of sloughing by applying a gentle lateral pressure).(2) Other clinical exam manifestations include pulmonic manifestations (pneumonia, interstitial pneumonitis and ARDS) and gastrointestinal manifestations (small bowel ulcerations, bowel perforation and intussusception).
In patients with severe skin detachment, complications in the acute setting include massive fluid loss through denuded skin, severe electrolyte abnormalities, hypothermia, hypovolemic shock, renal failure, insulin resistance, sepsis/septic shock (most often caused by Pseudomonas aeruginosa and Staphylococcus aureus) and multi-system organ failure.(2)
Thus, recognizing SJS/TEN and the other potentially life-threatening disease process becomes imperative. Among the differential includes, but not limited to:
- Pemphigus vulgaris (an autoimmune intraepithelial blistering disease on the skin and mucus membranes with tender, flaccid bullae and a positive Nikolsky’s sign)
- Necrotizing fasciitis (a severe flesh-eating and often polymicrobial soft tissue infection with a 100% mortality if not treated)
- Toxic shock syndrome (the result of a aureus exotoxin secondary to nasal packing, an abscess, or foreign body resulting in erythroderma, macular eruption, desquamation, a negative Nikolsky’s sign and multi-system organ failure)
- Staphylococcal scalded skin syndrome (the result of a S. aureus exotoxin resulting in diffuse erythroderma, exfoliation, fragile bulla, a positive Nikolsky’s sign, and no mucus membrane involvement)
- Classic and incomplete Kawasaki disease (febrile for at least five days with the addition of bilateral conjunctivitis, mucus membrane involvement, diffuse maculopapular rash, cervical adenopathy and changes to the extremities like erythema/swelling).
The diagnosis is considered a clinical diagnosis with no definitive imaging or lab abnormalities being diagnostic. Lab abnormalities may reveal an anemia, lymphopenia, neutropenia (poor prognosis), increased BUN, hyperglycemia, hypoalbuminemia and electrolyte imbalances; however, these are not diagnostic.(4)
Treatment
Treatment is multifaceted starting with an immediate diagnosis, evaluation of the severity of the illness, supportive treatment and early referral to a burn center.(3) These severe mucocutaneous adverse reactions are commonly the result of medication, as previously described, and as such, withdrawal of the offending agent is of paramount importance in improving the overall prognosis. Fluid and electrolyte abnormalities may be severe as a result of a hypercatabolic state and massive transdermal fluid loss.
Wound care often involves surgical debridement, moisture-retentive ointments, whirlpool therapy, anti-shear wound care or ophthalmic therapy as indicated.(1,4) Preventing and treating infections is important as these patients are at high risk of infection, with sepsis becoming a prominent cause of death. The majority of burn specialists do not advocate on prophylactic antibiotics, rather, management involves sterile handling, antiseptic solutions and obtaining cultures from skin/blood. If a patient clinically deteriorates, develops a fever, has a visible infection, and/or neutrophilia, then the initiation of prompt systemic antibiotics is recommended.(1)
Overall mortality is around 25%, with a morality of roughly 10% in SJS and 30% in patients with TEN.(1) The extent of cutaneous involvement is considered the main risk factor for morality within 90 days from disease onset. The most common causes of death within the hospital setting include: sepsis, acute respiratory distress syndrome and multi-system organ failure. A poor prognosis has been associated with elderly patients >70, human immunodeficiency virus, presence of renal failure, metastatic cancer and liver cirrhosis.(1,2)
References:
- High, W, Roujeau, J. “Stevens-Johnson syndrome and toxic epidermal necrolysis: Management, prognosis, and long-term sequelae.” May 25, 2018.
- High, W, Roujeau, J. “Stevens-Johnson syndrome and toxic epidermal necrolysis: Pathogenesis, clinical manifestations, and diagnosis.” August 16, 2018.
- Kosoko, A, Kaziny, B. “Recognize Pediatric Toxic Epidermal Necrolysis Symptoms, Manage Disease.” ACEP Now. February 11, 2015.
- Ferandiz-Pulido C, Garcia-Patos, V. A review of causes of Stevens-Johnson syndrome and toxic epidermal necrolysis in children. Arch Dis Child2013; 98:998.
- Patil, J, Motghare, V, Deshmukh, V, Jaykare, S, Pise, H. “Penicillin-Induced Stevens-Johnson Syndrome: A Case Report.” Indian Journal of Clinical Practice, Vol. 24, No. 7, December 2013.
- Beken, B, Can, C, Orencik, A, Can, N, Mehtap, Y. “Oxcarbazepine-induced Stevens-Johnson syndrome: a pediatric case report.” Oxford Medical Case Reports, 2017; 6, 79-81.
- Letko, E, Papaliodis, D, Papliodis, G, et al. Stevens-Johnson syndrome and toxic epidermal necrolysis: a review of the literature. Ann Allergy Asthma Immunol2005; 94:419.

1 Comment
nice bird eye view