Every shift, emergency physicians care for patients they don’t know in a time-pressured and overcrowded environment. Given that the majority of patients in the emergency department have non-life-threatening conditions, physicians are easily lulled into a false sense that the patient laying before them has a benign condition.
Avoid common errors by recognizing potentially-misleading indicators
Every shift, emergency physicians care for patients they don’t know in a time-pressured and overcrowded environment. Given that the majority of patients in the emergency department have non-life-threatening conditions, physicians are easily lulled into a false sense that the patient laying before them has a benign condition. The great challenge is to sort through the morass of benign conditions and properly diagnose and treat the deadly ones. On top of this, providers are expected to deliver excellent care efficiently, communicate with patients quickly, discharge them with proper outpatient care instructions, and then rapidly move on to the next case. Errors in this setting are inevitable, but you can minimize mistakes by learning to recognize and avoid certain common pitfalls.
Never assume that a recent negative stress test definitively rules out acute coronary syndrome
Many patients who present to the emergency department (ED) have already seen a physician for their chest pain (CP). Many have also had some form of provocative cardiovascular testing, an exercise electrocardiography stress test (EST), nuclear imaging, stress echocardiography, or even cardiac catheterization. There are pitfalls in relying too heavily on previous stress testing, and it is very important to know the limitations of this information.
Exercise electrocardiographic stress testing is common in the evaluation of low-risk patients presenting with CP and as an initial noninvasive strategy for evaluating CP after an appropriate evaluation to rule out acute myocardial infarction (AMI). ESTs use various protocols but share in common a methodology: an attempt to induce and measure cardiac ischemia. Sensitivity and specificity for ESTs are in the range of 68% and 77%, respectively, for single-vessel CAD. These numbers vary in each study, largely due to differences in the patient population being studied. Sensitivity and specificity also increase in the presence of multivessel disease. The prognosis for patients with a “negative EST” is reported to be good with 4-year survival rates as high as 98% to 99%.
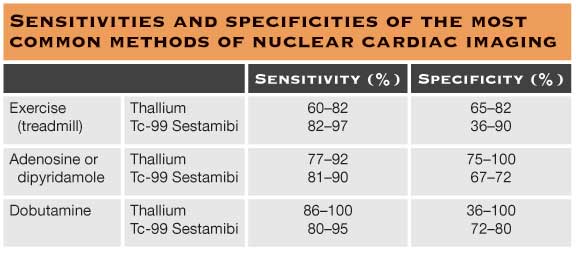
A logical extension of the EST is exercise or pharmacological stress nuclear imaging. There are now a variety of different protocols to image the heart using different radiotracers, both at rest and during exercise, in an attempt to unmask ischemia. Resting and exercise images are then compared to define nonperfusing areas of myocardium, reversible perfusion defects, and normal perfusion. Table 59.1 summarizes the sensitivities and specificities of the most common methods of nuclear cardiac imaging.
A highly positive nuclear study (one demonstrating large reversible perfusion defects) has been shown to predict a high annual event rate (death or MI) of 6% or greater. Patients with known CAD, diabetes mellitus, or a mildly abnormal nuclear study have event rate of 1% to 2% per year. However, a negative or normal cardiac nuclear imaging has a good prognosis with a <1% event rates per year.
Echocardiography can be used to detect wall motion abnormalities, which have been shown to correlate to areas of CAD. In isolation, echocardiography has an unacceptable high false-negative rate for ACS. Stress echocardiography, however, has demonstrated similar test characteristics to nuclear stress testing. The choice of one modality over another is usually a matter of what is available to the ordering clinician.
All forms of cardiac stress testing are designed to detect fixed coronary stenoses, not necessarily in patients at risk for ACS. Unfortunately, ACS frequently arises from unstable lesions that develop in plaques that cause a stenosis of 50% or less of the coronary lumen (so-called nonobstructive lesions). Although a negative stress test does statistically decrease the likelihood that a given patient will have an adverse event in the future, it does not directly assess for or predict the precursor to MI, which is an unstable intracoronary plaque.
Cardiac catheterization is ultimately the gold standard for the diagnosis of flow-limiting cardiovascular disease. So what if a patient with CP, who recently just had a cardiac catheterization, presents to the ED with CP—what risk stratification and prognostic information can the ED physician garner from a negative study or a minimally abnormal one? Recent studies from Italy and the United States have shown a completely normal coronary angiogram to portend an excellent prognosis with a negligible mortality at 10 years. However, other studies have found that a completely normal angiogram only had an event-free rate of 75% and a survival rate of 95% at 3 years. Patients who were at higher risk of events (defined as death, stroke, or reinfarction) were also more likely to be older, have diabetes or depressed ejection fractions. Other studies have suggested coagulopathies, cocaine abuse, and collagen vascular disease were present in MI with normal coronaries. These studies demonstrate that even with a completely normal coronary angiogram, there is still risk for MI.
So, what is the bottom line? When evaluating patients for ACS or AMI, do not rely too heavily on previous negative stress tests. Even a normal cardiac catheterization has its limitations.
Never rely on a single negative or indeterminate troponin to rule out acute coronary syndromes
Chest pain (CP) accounts for 6 million emergency department (ED) visits annually in the United States. Although missed diagnosis of acute myocardial infarction (AMI) has become less common, it remains an expensive source of malpractice suits filed against emergency physicians (EPs). Missed cases of unstable angina (UA), which may lead to MI and death, remain frustratingly common.
It has become commonplace to order an electrocardiogram (ECG), chest radiograph, and a serum troponin level on every patient presenting to the ED with CP. In fact, this is done in some cases by triage protocol before a physician even sees the patient. However, this reflexive ordering is not without risk.
Cardiac troponin T and I are released into circulation upon ischemic insult to myocardium. During AMI, troponin begins to rise within approximately 6 h and elevated levels are detectable for up to 2 weeks. Troponins are an important tool in evaluating and stratifying patients (both with and without CP) and have important implications with respect to mortality. Although assays differ, troponins have a well-defined range of sensitivity and specificity. Table 56.1 summarized from Balk et al.’s recent review summarizes the test characteristics of troponin T and I on initial presentation of CP and with serial measurements. Many studies have demonstrated the prognostic utility of detectable troponin levels upon presentation. One NEJM study demonstrated that 30-day mortality was significantly higher in patients presenting with AMI and a detectable troponin level. However, an undetectable troponin level does not rule out AMI. Troponin levels may take as many as 6 to 9 h to become detectable and sensitivity does not approach 100% until 12 h post cardiac event. Therefore, an early negative troponin (e.g., <6 h after symptom onset) necessitates a repe at troponin. The precise timing of the second troponin is currently the subject of much debate, and this may depend, in part, on the sensitivity of the specific assay used. Although some authors have suggested that two negative troponins separated by 2 h may be sufficient to rule out MI, others believe that longer intervals are necessary.
Many clinicians use the duration of CP to justify a single troponin “rule out.” For example, one might conclude that a single troponin rules out MI in patients in whom CP is >9 or more hours duration. However, it is also important to consider the nature of the symptoms. Ischemic CP may represent a number of different entities, ranging from stable angina to AMI. It may be difficult to distinguish at what point in time UA has become an AMI. For example, 12 h of CP may actually represent 11 h of UA and an MI that began in the past hour. Therefore, in most cases of CP, one troponin leads to another. A large study conducted in 12 American and Canadian EDs demonstrated that using CP duration alone as a criterion for obtaining a single cardiac marker will, in fact, result in missed AMIs.
UA is usually defined as new-onset angina, an increase in the frequency and/or duration of angina, or angina at rest. UA throws a bit of a wrinkle into algorithms for ruling out an ischemic source of CP because it only sometimes results in an elevation of cardiac biomarkers. Even with serial troponins, the sensitivity for UA is only 36%. This may be because ischemia that is sufficient to cause CP or discomfort may not be sufficient to compromise membrane integrity and result in troponin leak. Thus, even with serial negative troponins, there is truly no substitute for obtaining and documenting as accurate a history as possible. UA can be every bit as life threatening as AMI, and current AHA/ACC guidelines for the management of UA share many similarities with those for non-ST elevation MI. With regard to indeterminate range troponins, it is common for the EP to be told by an internist, “a troponin in the indeterminate range is negative for ACS.” However, this statement can be misleading and may result in poor outcomes for patients. The troponin is a single data point. One cannot predict in what direction the troponin will trend. Several studies have shown that patients presenting with indeterminate range troponins have a higher likelihood for adverse events, including AMI and need for invasive cardiac procedures.
So, what is the bottom line? A single troponin does not rule out acute coronary syndrome (ACS). If the decision to use biomarkers to evaluate a patient for MI is made, then at least two troponins spaced in time should be done. Secondly, UA may or may not be accompanied by a detectable troponin leak—go with the total clinical picture. Finally, indeterminate range troponins are misleading and a repeat troponin should be performed to ensure that the level is not rising.
Disclaimer
It is neither the purpose nor intent of this article to serve as a final authoritative source on any medical condition, treatment plan, or clinical intervention, nor should this article be used to rigorously define a rigid standard of care that should be practiced by all clinicians. Every attempt has been made to insure the accuracy of management recommendations and medication dosages. However, the reader is urged to consult other resources for confirmation of recommendations and medication dosages.
This article is an excerpt from the book “Avoiding Common Errors in the Emergency Department” (Lippincott Williams & Wilkins, 2010)



1 Comment
I had bad chest pains after riding a staionary bike,but I dissmissed it.The next day I felt somewhat dizzy and anxious with mild sweating.I felt something just wasn’t right.I called a cardiologists I had seen years before but he was out of the office for a week.His nurse told me to go to the er.I went to the er closes to me which is not where they practice.They hooked me up to an ekg and then a monitor.The ekg was normal.They unhooked the monitor so i could use the rest room and never came back to hook it up until they decided to admit me on the grounds that my troponin I was elavated at0.11.During the night my levels rose to0.15 and then 0.19.I was told that I would be having a stress test in the morning.I also had an attack that night where my arems were shaking my hands were tingling and I was very disoriented,
but they said my ekg was normal.The next morning I was awokened and told that my levels were back down to where they had syarted0.11 and I was being released.I was shocked.What do you think.I’m 48yrs old and was in great shape.I do have a lot of vascular and heart disease in my family.Both my father and his sister died very young from it.Any thoughts?